Find verified Ondansetron API manufacturers, suppliers, and distributors across the globe. Explore companies that offer high-purity Ondansetron for pharmaceutical use, along with product details, certifications, and reliable sourcing options. Compare prices, check availability, and connect directly with trusted suppliers for Ondansetron API.
Alternate Names: Zudan, Zuplenz, Zofran ODT
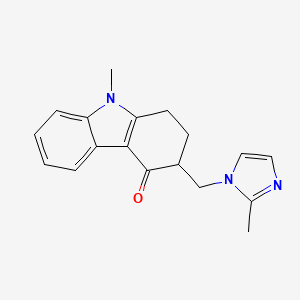
CAS No: 99614-02-5
PubChem CID: 4595
Mol Formula: C18H19N3O
Mol Weight: 293.4 g/mol
IUPAC Name: 9-methyl-3-[(2-methylimidazol-1-yl)methyl]-2,3-dihydro-1H-carbazol-4-one
API Description: A competitive serotonin type 3 receptor antagonist. It is effective in the treatment of nausea and vomiting caused by cytotoxic chemotherapy drugs, including cisplatin, and has reported anxiolytic and neuroleptic properties. Having been developed in the 1980s by GlaxoSmithKline and approved by the US FDA since January 1991, ondansetron has demonstrated a long history of use and efficacy. Commonly formulated as oral tablets, orally disintegrating tablets (ODT), and injections, and available as generic products as well, ondansetron continues to see contemporary innovations in its formulation and use, including the development of orally soluble films that are both discreet in administration and less of a burden in comparison to having patients attempt to swallow pills during emesis. The FDA withdrew its approval for the use of all intravenous drug products containing more than 16 mg of ondansetron hydrochloride in a single dose, due to a high risk of QT prolongation.