Find verified Asciminib API manufacturers, suppliers, and distributors across the globe. Explore companies that offer high-purity Asciminib for pharmaceutical use, along with product details, certifications, and reliable sourcing options. Compare prices, check availability, and connect directly with trusted suppliers for Asciminib API.
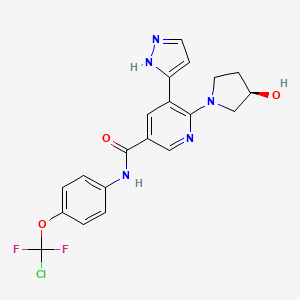
Alternate Names: Asciminib free base, asciminibum
CAS No: 1492952-76-7
PubChem CID: 72165228
Mol Formula: C20H18ClF2N5O3
Mol Weight: 449.8 g/mol
IUPAC Name: N-[4-[chloro(difluoro)methoxy]phenyl]-6-[(3R)-3-hydroxypyrrolidin-1-yl]-5-(1H-pyrazol-5-yl)pyridine-3-carboxamide
API Description: Asciminib is a tyrosine kinase inhibitor (TKI) used in the treatment of chronic-phase Philadelphia chromosome-positive chronic myeloid leukemia (Ph+ CML). More specifically, it is an inhibitor of the ABL1 kinase activity of the BCR-ABL1 fusion protein which serves as a driver of CML proliferation in most patients with the disease. It has also shown benefit in Ph+ CML with the T315I mutation, which produces a mutant BCR-ABL1 which is typically treatment-resistant as compared to wild-type BCR-ABL1. Existing inhibitors of ABL compete at the ATP binding sites of these proteins and can be classified into those that target the active conformation of the kinase domain ([dasatinib], [bosutinib]) and those that target the inactive kinase domain ([imatinib], [nilotinib], [ponatinib]). Asciminib is unique in that it acts as an allosteric inhibitor, binding at the myristoyl pocket of the BCR-ABL1 protein and locking it into an inactive conformation. Asciminib received FDA approval on October 29, 2021 (Scemblix, Novartis AG).