Find verified Selexipag API manufacturers, suppliers, and distributors across the globe. Explore companies that offer high-purity Selexipag for pharmaceutical use, along with product details, certifications, and reliable sourcing options. Compare prices, check availability, and connect directly with trusted suppliers for Selexipag API.
Alternate Names: Uptravi, ACT-293987
CAS No: 475086-01-2
PubChem CID: 9913767
Mol Formula: C26H32N4O4S
Mol Weight: 496.6 g/mol
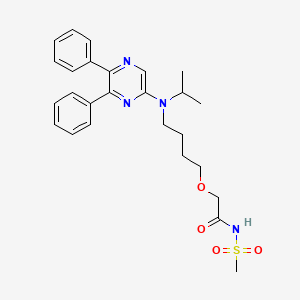
IUPAC Name: 2-[4-[(5,6-diphenylpyrazin-2-yl)-propan-2-ylamino]butoxy]-N-methylsulfonylacetamide
API Description: Selexipag was approved by the United States FDA on December 22, 2015 for the treatment of pulmonary arterial hypertension (PAH) to delay disease progression and reduce risk of hospitalization. PAH is a relatively rare disease with usually a poor prognosis requiring more treatment options to prolong long-term outcomes. Marketed by Actelion Pharmaceuticals under brand name Uptravi, selexipag and its active metabolite, ACT-333679 (MRE-269), act as agonists of the prostacyclin receptor to increase vasodilation in the pulmonary circulation and decrease elevated pressure in the blood vessels supplying blood to the lungs.