Find verified Roflumilast API manufacturers, suppliers, and distributors across the globe. Explore companies that offer high-purity Roflumilast for pharmaceutical use, along with product details, certifications, and reliable sourcing options. Compare prices, check availability, and connect directly with trusted suppliers for Roflumilast API.
Alternate Names: Daliresp, BY-217, ZORYVE, DAXAS, roflumilastum
CAS No: 162401-32-3
PubChem CID: 449193
Mol Formula: C17H14Cl2F2N2O3
Mol Weight: 403.2 g/mol
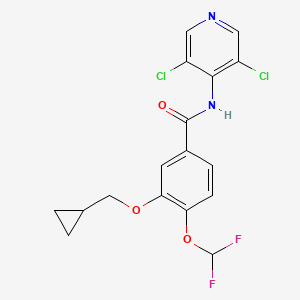
IUPAC Name: 3-(cyclopropylmethoxy)-N-(3,5-dichloropyridin-4-yl)-4-(difluoromethoxy)benzamide
API Description: Roflumilast is a highly selective phosphodiesterase-4 (PDE4) inhibitor. PDE4 is a major cyclic-3',5′-adenosinemonophosphate (cyclic AMP, cAMP)-metabolizing enzyme expressed on nearly all immune and pro-inflammatory cells, in addition to structural cells like those of the smooth muscle or epithelium. The resultant increase in intracellular cAMP induced by roflumilast's inhibition of PDE4 is thought to mediate its disease-modifying effects, although its precise mechanism of action has yet to be elucidated. The oral formulation of roflumilast is indicated to manage chronic obstructive pulmonary disease. It was first approved by the EMA in July 2010, and by the FDA in January 2018. Roflumilast topical cream is indicated to treat plaque psoriasis. The cream formulation was first approved by the FDA in July 2022 and by Health Canada in April 2023. On December 15, 2023, the FDA approved a new topical foam formulation of roflumilast for the treatment of seborrheic dermatitis in patients aged 9 years and older.